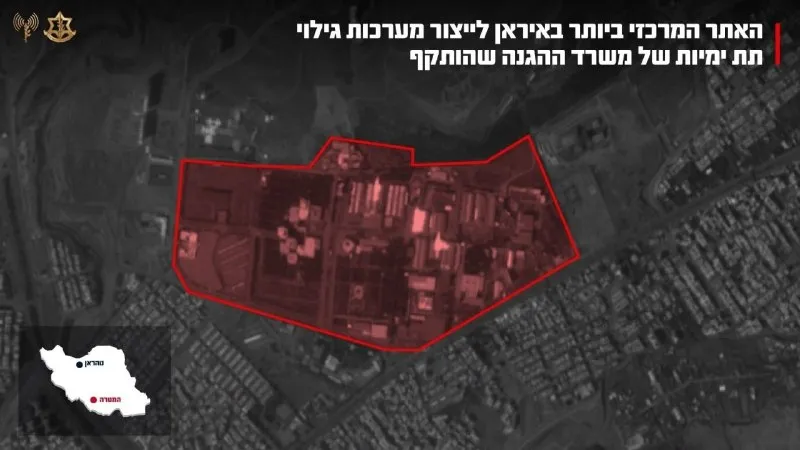
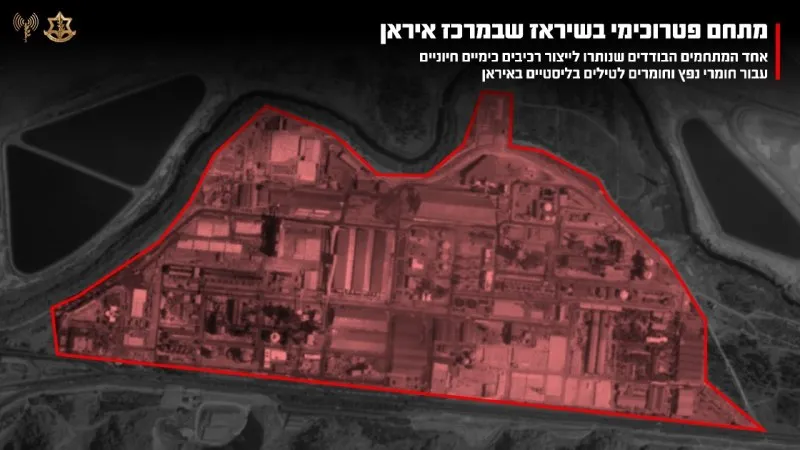
Shirkadda Israa’iili ee Tignoolajiyada Caafimaadka ee IceCure Oo Helaysa Ogolaanshaha FDA Daraasad Dambe Oo Ka-dambeysa Kansarka Naasaha
Daaweynta kansarka naasaha ee IceCure Medical ee ProSense cryoablation, oo ah daaweyn aan qalliin lahayn, ayaa hadda la baarayaa FDA-da iyadoo la sameynayo daraasad la yiraahdo "ChoICE.
Yeruusaalem, 12 Maarso, 2026 (TPS-IL) — Shirkadda Israa‘iili ee farsamada caafimaadka ee IceCure Medical ayaa helay ogolaansho ka timid FDA-da si ay u fuliso daraasad ka dib suuq-geyn ah oo lagu magacaabo “ChoICE” oo ku saabsan daaweynta ProSense cryoablation, oo ah daaweyn aan qalliin lahayn oo lagu daweeyo kansarka naasaha ee khatarta yar. Daraasadda oo ka dhici doonta Mareykanka ayaa la filayaa inay ku biiraan ilaa 400 bukaan oo ku kala yaal 30 goobood oo caafimaad muddo saddex sano ah, waxaana la filayaa in ugu yaraan 80 bukaan la arko sanadka ugu horreeya.
ProSense waxa la ansixiyay Oktoobar 2025, waxaana loo isticmaali karaa bukaanada da’doodu tahay 70 sano ama ka weyn, kuwaas oo qaba burooyinka cabbirkoodu yahay 1.5 cm ama ka yar. Kadib ogolaanshahaas, FDA-da ayaa codsatay daraasad ka dib suuq-geyn ah si loo helo xog dhab ah oo ku saabsan isticmaalka daaweynta. Daraasadda “ChoICE” oo ka dhici doonta Mareykanka ayaa la filayaa inay ku biiraan ilaa 400 bukaan oo ku kala yaal 30 goobood oo caafimaad muddo saddex sano ah, waxaana la filayaa in ugu yaraan 80 bukaan la arko sanadka ugu horreeya. Goobaha ka qayb-qaadanaya waxay sidoo kale ku daweyn karaan bukaanada dheeraadka ah ganacsi ahaan, iyadoo la siinayo koodka dib-u-bixinta ee CPT Category III.